DFG Priority Program SPP 2080
"Catalysts and reactors under dynamic conditions for energy storage and conversion"
Chemical storage of renewable energy
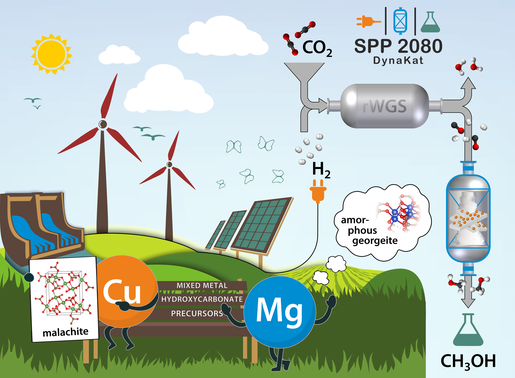
as example reaction for SPP2080. Copyright: Gereon Behrendt/ChemCatChem.
The fluctuating availability of renewable energies such as wind and solar power represents one of the greatest challenges in the context of the energy transition. Electricity generated on windy and sunny days can be stored in the form of chemical energy carriers such as hydrogen or hydrocarbons. This requires the use of catalysts, reactors and electrochemical cells under externally controlled dynamic reaction conditions. However, the influence of dynamic conditions on catalytic reaction systems has hardly been considered so far, as chemical reactors are mostly operated in stationary mode.
Catalysts under dynamic conditions
Recent investigations have revealed that the structure of solid catalysts and thus also the catalytic effect can change considerably with the reaction conditions. On the one hand, there is the potential to increase the yield of the desired reaction products through dynamic operation and to reactivate catalysts in rest phases. On the other hand, the nanostructured catalysts must be stabilized. Efficient utilization of dynamic reaction conditions therefore requires a fundamental understanding of all the processes involved - from the atomic scale of the catalyst to three-dimensional concentration and temperature distributions in the technical reactor. The rapid development of spectroscopic methods and modelling combined with new approaches in material and reactor design offer excellent prerequisites for this.
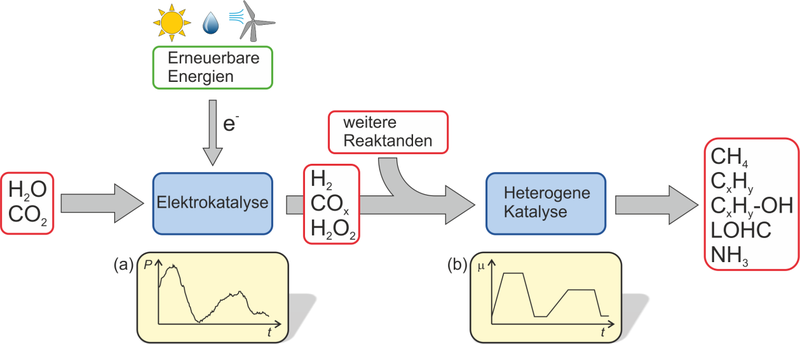
This is where the Priority Programme 2080 steps in. Applied to current questions of energy transition, a basic understanding of microscopic processes on solid catalysts under dynamic conditions and their effects on activity, selectivity and stability will be developed. The transport processes on the mesoscale, which are important for the behaviour of technical catalysts, will also be considered. The interdisciplinary research program is located in Technical Chemistry or Chemical Reaction Engineering and is explicitly open to other areas of chemistry, physics or materials science that contribute to the topic.
Interdisciplinary consortia
In interdisciplinary consortia, fundamental and methodological challenges of dynamic operation are examined in five closely linked subject areas:
(A) Characterisation using "operando" methods, i.e. under reaction conditions to understand solid catalysts "at work" under dynamic conditions,
(B) Predictive theoretical description of active phases forming under dynamic reaction conditions and elementary steps occurring at the catalyst,
(C) Kinetics and multiscale modelling involving atomic information to understand the behaviour of catalysts and electrodes under technical and dynamic conditions,
(D) Targeted materials design to stabilize catalytically active surface structures and to investigate them under periodic reaction conditions,
(E) Novel reactor and electrolyser concepts for methodical investigations under transient conditions.
Efficient operation
The expected increase in knowledge should enable the efficient operation of catalytic processes under dynamic reaction conditions. Its basic understanding will be developed using the example of reactions to energy storage and conversion (Fig. 1) and creates the basis for future technical applications. It is characteristic for all investigated systems that the dynamics are systematically imposed from the outside in the time domain between seconds and days, either because the applied dynamics can only be avoided with great effort (e.g. fluctuating supply of electrical energy), or because justified advantages for space-time yields or selectivities of the catalytic reactions are expected from the dynamic operation. The expected increase in knowledge is also interesting for other areas such as exhaust gas catalysis, selective oxidations, fuel cells, batteries or photocatalysis. However, these applications, as well as purely physico-chemical studies, are not the focus of this SPP.
[1] K. F. Kalz, R. Kraehnert, M. Dvoyashkin, R. Dittmeyer, R. Gläser, U. Krewer, K. Reuter, J.-D. Grunwaldt, ChemCatChem 2017, 9, 17-29.
