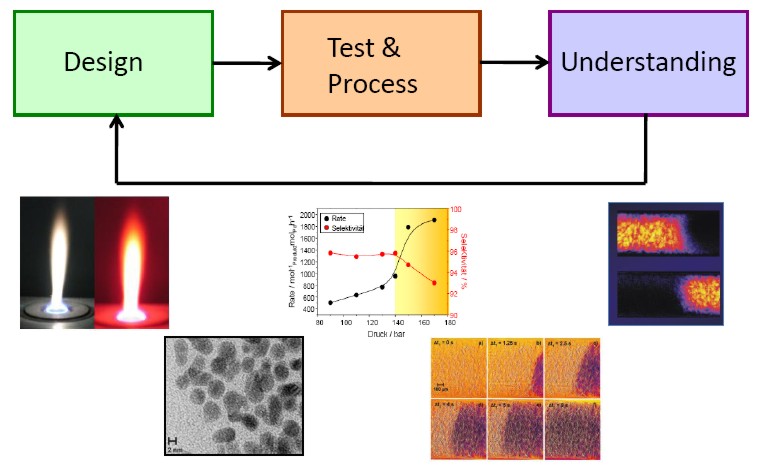
Our Research focuses on the development of new catalysts within biomass conversion, fine chemistry and catalysis for emission control. More than 60% of all chemical processes are based on catalytic processes among which heterogeneous catalysts play and will play the main role in bulk chemicals, also during the present change from fossile to renewable energies. Thereby three subject are of paramount importance:
(I) Design: synthesis of new nanomaterials
(II) Test & process: efficient catalyst testing
(III) Understanding: fundamental understanding using physico-chemical analysis and in situ spectroscopy.
This combined approach is used for a rational design and improvement of catalytic processes.
We are looking for motivated students for bachelor/master theses! The broad knowledge in the field of catalysis that can be acquired while working on one of our projects will benefit any student for furthering her/his career towards industry or academia. You can find short abstracts on possible bachelor/master theses below the research topics or directly contact us!
Topics:
- Nanomaterials: New preparation Techniques and High Throughput Testing
- Environmental Catalysis
- Energy-related Catalysis
- Biomass Conversion
- Selective Oxidation Reactions
- In situ / operando Characterization of Heterogeneous Catalysts
Catalyst and Nanomaterial Preparation
Nanomaterials: New preparation Techniques and High Throughput Testing
Novel preparation methods to new nanomaterials, efficient catalytic tests and proper catalyst characterization are our basis for the design of new catalysts. For example, currently applied synthesis strategies include flame synthesis, preparation of size-controlled particles or hydrothermal synthesis.
The Target Reactions
Environmental Catalysis
Environmental pollution has been a global matter of concern for many years, leading to progressively stricter regulations. Particularly, air contamination by emitted NOx, CO, CO2, CH4, volatile organic compounds and particulate matter is regarded as a major problem of our society with harmful effects on human health and environment. In addition to minimizing the emissions directly at the source, e.g. reducing the fuel consumption of a combustion engine, the catalytic aftertreatment of the emitted pollutants is regarded as one of the most promising approaches.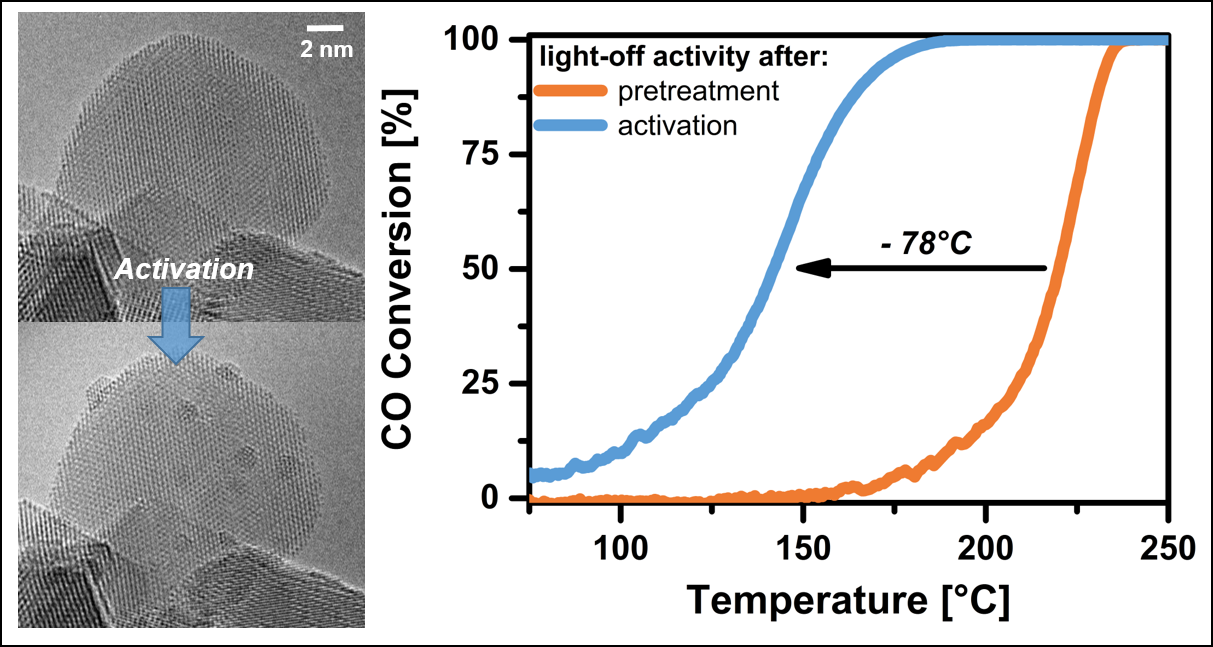 This strategy applies to both, mobile and stationary emission sources from industry, transportation to household.
This strategy applies to both, mobile and stationary emission sources from industry, transportation to household.
To comply with the more stringent emission laws a continuous development of new and improved generation of emission control catalysts is required. In this context, our group is conducting numerous studies on different classes of catalysts applied in the aftertreatment system of diesel, gasoline and natural gas engines as well as for reducing household emissions. By fundamental understanding of catalyst structural and mechanistic aspects, we aim at improving its efficiency and durability. For example, we were able to significantly enhance the catalytic performance by directly acting on the noble metal particle size in a Pt-CeO2-based catalyst. In collaboration with our industrial partners, we observed that short reducing pulses lead to a slight increase of Pt particles at mild temperature, which has a tremendous effect on the catalyst activity.
For more details, have a look at the Emission Control Center with Prof. Olaf Deutschmann.
Possible bachelor/master thesis: topic 1
Related publications:
A. M. Gänzler, M. Casapu, D. E. Doronkin, F. Maurer, P. Lott, P. Glatzel, M. Votsmeier, O. Deutschmann O, J. -D. Grunwaldt, J. Phys. Chem. Lett. 2019, 10(24), 7698.
A. M. Gänzler, M. Casapu, P. Vernoux, S. Loridant, F. J. Cadete Santos Aires, T. Epicier, B. Betz, R. Hoyer, J.-D. Grunwaldt, Angew. Chem. Int. Ed. 2017, 56, 13078.
A. Gremminger, P. Lott, M. Merts, M. Casapu, J. -D. Grunwaldt, O. Deutschmann, Appl. Catal. B 2017, 218, 833.
Energy-related Catalysis
Climate change and the global energy demands have led to increased efforts on efficient renewable energy storage aiming at a CO2 neutral approach. Closely related to this is the production of green hydrogen and synthesis gas (CO/H2), thermochemically or electrochemically. Our contribution in this direction is state-of-the-art research on catalyst development, process optimization and fundamental understanting.
Power to You via Power2X - Synthesis of energy storage materials from CO and CO2
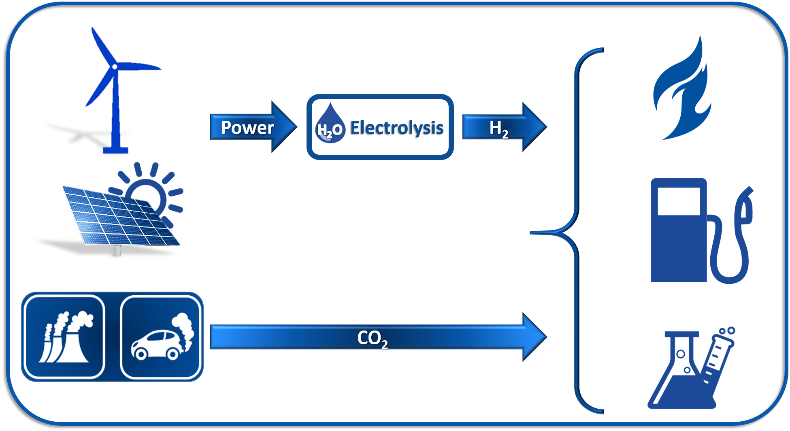 Recent advances in renewable wind and solar energy generation have made this a viable long-term option for fulfilling the increasing energy needs of our society. There is, however, a substantial imbalance between the power production and its utilization capacity. In order to circumvent these issues, efficient energy storage solutions need to be developed. One example is the Power-to-X strategy, at the core of which is the conversion of CO/CO2 and H2 gas mixtures into fuels and intermediate chemicals. In this framework, our group deals with several aspects of the Power2X strategy, including water electrolysis, methanation, FTS, methanol and higher alcohols synthesis from CO/CO2+H2. The expertize we aim to bring into this global challenge is determining the catalyst behaviour under dynamic conditions that mimic nature’s character using advanced techniques, such as in-situ and operando XAS, EXAFS, etc.
Recent advances in renewable wind and solar energy generation have made this a viable long-term option for fulfilling the increasing energy needs of our society. There is, however, a substantial imbalance between the power production and its utilization capacity. In order to circumvent these issues, efficient energy storage solutions need to be developed. One example is the Power-to-X strategy, at the core of which is the conversion of CO/CO2 and H2 gas mixtures into fuels and intermediate chemicals. In this framework, our group deals with several aspects of the Power2X strategy, including water electrolysis, methanation, FTS, methanol and higher alcohols synthesis from CO/CO2+H2. The expertize we aim to bring into this global challenge is determining the catalyst behaviour under dynamic conditions that mimic nature’s character using advanced techniques, such as in-situ and operando XAS, EXAFS, etc.
Bachelor and master thesis available!
Related publications
J. Geppert, P. Röse, S. Czioska, D. Escalera-López, A. Boubnov, E. Saraçi, S. Cherevko, J.-D. Grunwaldt, U. Krewer. J. Am. Chem. Soc. 2022, 144, 29.
Pandit, L.; Serrer, M.-A.; Saraҫi E.; Boubnov, A.; Grunwaldt, J.-D. Chem. Methods. 2022, 2, e202100078.
Pandit, L.; Boubnov, A.; Behrendt, G.; Mockenhaupt, B.; Chowdhury, C.; Jelic, J.; Hansen, A.-L.; Saraçi, E.; Ras, E.-J.; Behrens, M.; Studt, F.; Grunwaldt, J.-D. ChemCatChem, 2021, 13, 4120.
Czioska, S.; Boubnov, A.; Escalera-López, D.; Geppert, J.; Zagalskaya, A.; Röse, P.; Saraçi, E.; Alexandrov, V.; Krewer, U.; Cherevko, S.; Grunwaldt, J.-D. ACS catalysis, 2021, 11, 10043.
Escalera-López, D.; Czioska, S.; Geppert, J.; Boubnov, A.; Röse, P.; Saraçi, E.; Krewer, U.; Grunwaldt, J.-D.; Cherevko, S. ACS catalysis, 2021, 11, 9300.
Serrer, M.-A.; Stehle, M.; Schulte, M. L.; Besser, H.; Pfleging, W.; Saraçi, E.; Grunwaldt, J.-D. ChemCatChem, 2021, 13, 3010.
Serrer, M.-A.; Gaur, A.; Jelic, J.; Weber, S.; Fritsch, C.; Clark, A. H.; Saraçi, E.; Studt, F.; Grunwaldt, J.-D. Catal. Sci. Techn. 2020, 10, 7542.
Loewert, M.; Serrer, M.-A.; Carambia, T.; Stehle, M.; Zimina, A.; Kalz, K.; Lichtenberg, H.; Saraçi, E.; Pfeifer, P.; Grunwaldt, J.-D. React. Chem. Eng. 2020, 5, 1071.
M.-A. Serrer, K. F. Kalz, E. Saraҫi, H. Lichtenberg, J.-D. Grunwaldt. ChemCatChem, 2019, 11, 5018.
K. M. Walter, M.-A. Serrer, W. Kleist, J.-D. Grunwaldt. 2019. Appl. Catal. A, 2019, 585, 117150.
Biomass Conversion
Biomass is a renewable and abundant carbon-neutral resource for sustainable production of chemicals, among which fuels, monomers and other chemical building blocks. Our research starts with fundamental studies on the conversion of biomass platform molecules, e.g. fructose/glucose, HMF, and fatty acids, as well as catalytic upgrading of unpurified bio-derived solutions. Important reactions are oxidation, cycloaddition, oxidative cleavage, hydrogenation, etc. Comprehensive studies including catalyst preparation, screening, advanced characterization and reaction engineering, provide rationallity in catalyst design and process optimization for a sustainable future.
Growing Plant Bottles in the Lab - Bio-derived polymer building blocks for green bottles via innovative catalysis
 The synthesis of polymer building units from biomass is an attractive alternative to petrochemicals addressing issues such as depleting resources and sustainability.
The synthesis of polymer building units from biomass is an attractive alternative to petrochemicals addressing issues such as depleting resources and sustainability.
The packaging industry, and more specifically the production of plastic bottles, has the highest market demand and is majorly affected by the lack of fossil aromatics and the environmental regulations. Thus, the synthesis of polyethylene terephthalate (PET), or polyethylene furanoate (PEF), building blocks from renewable resources is an important objective in science and industry. In our group, different innovative routes are being developed in order to effectively synthesize these monomers from biomass. Catalyst preparation, characterization and reaction kinetics are the tools used to optimize the process and obtain a green product.
Bachelor and master thesis available!
Related publications
Neukum, D.; Baumgarten, L.; Wüst, D.; Sarma, B. B.; Saraçi, E.; Kruse, A.; Grunwaldt, J.-D. ChemSusChem. 2022,15, e2022004.
O.R. Schade, F. Stein, S. Reichenberger, A. Gaur, E. Saraci, S. Barcikowski, J.-D. Grunwaldt. Adv. Synth. Catal. 2020, 362, 5681
W. Naim, O.R. Schade, E. Saraci, D. Wüst, A. Kruse, J.-D. Grunwaldt. ACS Sust. Chem. Eng. 2020, 8, 11512
Schade, O. R.; Gaur, A.; Zimina, A.; Saraci, E.; Grunwaldt, J.-D. Catal. Sci. Techn. 2020, 10, 5036.
Schade, O.; Dolcet, P.; Nefedov, A.; Huang, X.; Saraçi, E.; Wöll, C.; Grunwaldt, J.-D.
Catalysts, 2020, 10, 342.
Selective Oxidation Reactions
Selective Oxidation of Propylene
 Selective oxidation processes are very important reactions in chemical industry for the production of myriad intermediates. The oxidation of propylene to acrolein is one of these selective oxidation reactions. Acrolein is further on used in different fields e.g. for the production of acrylic acid, an important polymer building block. Today, multicomponent metal oxide catalysts based on bismuth molybdates are used in the oxidation process to ideally achieve a high propylene conversion combined with a high acrolein selectivity.
Selective oxidation processes are very important reactions in chemical industry for the production of myriad intermediates. The oxidation of propylene to acrolein is one of these selective oxidation reactions. Acrolein is further on used in different fields e.g. for the production of acrylic acid, an important polymer building block. Today, multicomponent metal oxide catalysts based on bismuth molybdates are used in the oxidation process to ideally achieve a high propylene conversion combined with a high acrolein selectivity.
We extensively studied the basic system of bismuth molybdates and could recently reveal a structure activity/selectivity relationship by combining different characterization methods like X-ray diffraction (XRD), X-ray absorption spectroscopy (XAS) and Raman spectroscopy. Additionally, we are also investigating multicomponent systems, which represent the next step on the way from fundamental studies to real reactor ensembles. These systems consist of Bi-Mo-Co-Fe oxides and therefore numerous phases are present.
All the steps in catalyst research - namely preparation, characterization and testing - are performed within our group. The catalysts are prepared by flame spray pyrolysis and the catalytic performance is determined in a self-build setup. For characterization, we typically use Raman spectroscopy, XRD and nitrogen physisorption. Selected samples are subsequently studied by further techniques and in situ/operando to gain more insights into those systems and therefore to deepen our knowledge. By the combination of preparation, characterization and testing, we are able to rationally design and improve our systems based on the previously obtained results.
Related publications
P. Sprenger, W. Kleist, J.-D. Grunwaldt, ACS Catal. 2017, 7, 5628-5642.
P. Sprenger, M. Stehle, A. Gaur, A. M. Gänzler, D. Gashnikova, W. Kleist, J.-D. Grunwaldt, ACS Catal. 2018, 8, 6462-6475.
P. Sprenger, T. Sheppard, J.-P. Suuronen, A. Gaur, F. Benzi, J.-D. Grunwaldt, Catalysts 2018, 8, 356.
The Fundamental Understanding
In situ / operando Characterization of Heterogeneous Catalysts
Catalysts are ubiquitous in industry for improving the efficiency and sustainability of chemical reactions. Since catalyst structure and function are inseparable, it is essential to develop a level of understanding and control over the so-called ‘structure-activity’ relationships of a given catalyst system. With this in mind, the most accurate and realistic catalyst characterisation results rely on ‘in situ’ and ‘operando’ analysis.
- In situ means the application of analytical methods such as spectroscopy, diffraction or microscopy, under conditions relevant for catalytic function (e.g. reactive gases, temperature, pressure; these can be model or real conditions).
- Operando means the simultaneous collection of catalytic activity data, together with in situ measurements (making operando a special class of in situ).
Heterogeneous catalysts are complex materials which come in many shapes and sizes, often exhibiting structural elements and features of interest on multiple length scales. These can range from metal active sites (atomic to nm scale), to porosity (micro-/meso-/macropores on nm to µm scale), to global catalyst form or morphology (mm to cm scale). Multimodal and multiscale characterisation is therefore essential to develop full understanding of catalyst form and function. This includes surface methods such as XPS and DRIFTS (IR) to bulk analytical methods such as XRD and Raman. Synchrotron radiation plays a key role in this, with techniques ranging from X-ray absorption spectroscopy (XAS), to photon-in photon out spectroscopies (XES), to high resolution synchrotron X-ray diffraction (XRD), and imaging studies using X-ray microscopy.
In our group, in situ and operando characterisation is generally divided into three strongly interacting areas:
(i) design and construction of in situ / operando reactors and cells
(ii) application of in situ and operando spectroscopy, diffraction and microscopy
(iii) correlative characterization by multiple complementary techniques (e.g. combination of XAS, XRD, and Raman, or XAS and DRIFTS).
A deep understanding of catalytic performance can only be obtained through deriving structure-activity relationships. This in turn leads to the rational design of more efficient catalysts tuned for specific reactions and chemical processes. These activities strongly interact with industry, where real catalysts are applied.
Measurements can be performed at the CAT-ACT beamline, a new beamline dedicated to CATalysis and ACTinide research at the KIT synchrotron!
Looking at the Bigger Picture - Combining hard X-ray and electron microscopy for correlative catalyst imaging
 Imaging techniques play a fundamental role in catalyst characterisation. They provide information on sample structure, composition and morphology with high spatial resolution and at different length scales.
Imaging techniques play a fundamental role in catalyst characterisation. They provide information on sample structure, composition and morphology with high spatial resolution and at different length scales.
Transmission electron microscopy (TEM) is optimal for high resolution imaging down to atomic scale, but with strictly limited environmental conditions like gas and temperature. The sample may also be compromised by the need to prepare nanometre thin specimens, and the use of high energy electrons for imaging. On the other hand, hard X-ray microscopy (XRM) performed at synchrotron radiation sources can flexibly operate at elevated pressures or temperatures, and with micrometre to millimetre thick samples. Compared to electrons, hard X-rays are relatively non-invasive to the sample. However, spatial resolution is currently limited to some tens of nanometres. In reality, these techniques are highly complementary and the best of both methods can be combined using appropriate sample environments. XRM and TEM can provide not only imaging results, but also spectroscopic and diffraction data for a detailed understanding of the sample. One focal point is development of ‘in situ’ reactors, allowing catalyst imaging under relevant reaction conditions - the optimal way to observe catalysts at work and develop structure-activity relationships. Harnessing the power of these methods is a key research goal in the group.
Related publications
Y. Fam, T.L. Sheppard, A. Diaz, T. Scherer, M. Holler, W. Wang, D. Wang, P. Brenner, A. Wittstock, J.-D. Grunwaldt, ChemCatChem 2018, 10, 2858
S. Baier, C.D. Damsgaard, M. Klumpp, J. Reinhardt, T. Sheppard, Z. Balogh, T. Kasama, F. Benzi, J.B. Wagner, W. Schwieger, C.G. Schroer, J.-D. Grunwaldt, Microsc. Microanal. 2017, 23, 501.
S. Baier, A. Wittstock, C.D. Damsgaard, A. Diaz, J. Reinhardt, F. Benzi, J. Shi, T. Scherer, D. Wang, C. Kübel, C.G. Schroer, J.-D. Grunwaldt, RSC Adv. 2016, 6, 83031
Seeing is Believing - 3D catalyst characterization with harx X-ray tomography at the synchrotron

Heterogeneous catalysts are hierarchically-structured materials, which often show complex structural elements on multiple length scales. This includes metal nanoparticles or clusters, porosity (including micro-, meso- and macropores), and global structure or morphology, such as honeycombs, pellets or layers. Detailed characterisation of all these features and on all length scales is essential to develop understanding of catalyst form and function.
Hard X-ray micro- and nanotomography offers a versatile toolbox to meet this challenge. By taking advantage of the high flux, tuneable energy and sensitive detection capabilities of modern synchrotron radiation sources, it is possible to monitor the structure of catalysts in 3D. This can be combined with numerous contrast modes, including absorption (XAS), fluorescence (XRF), diffraction (XRD) or developing coherent methods such as ptychography. Furthermore, development of new sample environments even now permits 3D imaging in situ - under reaction conditions. These powerful methods are exploited in our group to look at everything from distribution and behaviour of metal nanoparticles, mapping of pore networks, and monitoring common catalyst deactivation processes such as sintering or coking. The many unexplored possibilities will surely lead to a bright future for X-ray tomography in catalysis research.
Related publications
P. Sprenger, T.L. Sheppard, J.-P. Suuronen, A. Gaur, F. Benzi, J.-D. Grunwaldt, Catalysts 2018, 8, 356
T.L. Sheppard, S.W.T. Price, F. Benzi, S. Baier, M. Klumpp, R. Dittmeyer, W. Schwieger, J.-D. Grunwaldt, J. Am. Chem. Soc. 2017, 139, 7855
G. Hofmann, A. Rochet, E. Ogel, M. Casapu, S. Ritter, M. Ogurreck, J.-D. Grunwaldt, RSC Adv. 2015, 5, 6893
CATACT-beamline at KIT-Lightsource
CATACT is part of the KIT Light Source. It is is an electron synchrotron at the Karlsruhe Institute of Technology (KIT), which serves as a synchrotron radiation source. Together with KIT, the KIT-Lightsource belongs to the Helmholtz Association of German Research Centers.
The CAT end station of the CATACT beamline at KIT Light Source is dedicated to catalytic research using X-ray spectroscopic techniques. It is operated by the Grunwaldt group at the Institute of Catalysis Research and Technology ( IKFT) and Institute for Chemical Technology and Polymer Chemistry (ITCP).
