Open positions
We offering a position for a master thesis on the topic: Oxidative Cleavage of C-C Bonds using Zeolite Catalysts
Catalysis for Sustainable Chemicals and Energy - Cat4Sus group
Catalyst and process development for upgrading renewable feedstock (biomass and waste) into commodity chemicals is an important topic in the group.
Cutting-edge research is also dedicated to understanding the catalysts and process dynamics in the hydrogenation of CO/CO2 via thermal and electrocatalytic pathways to energy dense molecules (alcohols and hydrocarbons).
Green H2 production via electrocatalytic splitting of water is also investigated with advanced operando methods.
Catalysis for Sustainable Chemicals and Energy - Cat4Sus group
↵
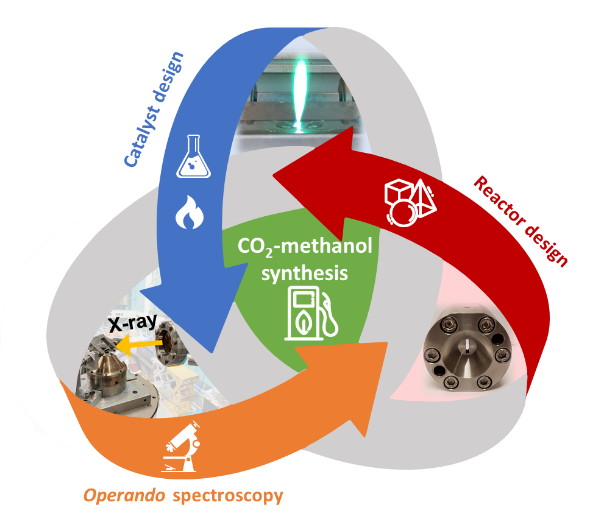
Methanol Synthesis from CO2
To lower CO2 emissions and gain energy independence, the amount of energy produced by renewable resources like wind and solar is increasing. Fluctuating weather and seasonal circumstances require the possibility to store surplus energy, so it can be used even if the energy production is lowered due to natural conditions. Within the concept of Power-to-Liquid, green hydrogen produced by water electrolysis powered by renewable energy, and CO2 from the atmosphere are combined to form a liquid energy carrier, for example methanol. The synthesis of methanol from syngas (CO+H2 + 3-5% CO2) is a well-studied and applied process which requires an active catalyst. However, for a CO2-based methanol production new challenges arise e.g. catalyst deactivation due to higher water concentration and unfavorable thermodynamics in comparison to syngas feed. Therefore, catalyst- and process optimization is necessary. Within our research, we use flame spray pyrolysis and other techniques for catalyst synthesis, design and develop cells/reactors for spectroscopic applications e.g. X-ray spectroscopy with the aim to perform operando and in situ studies under realistic CO2 methanol conditions.
PhD students: M. Schulte, L. Baumgarten
Thesis available
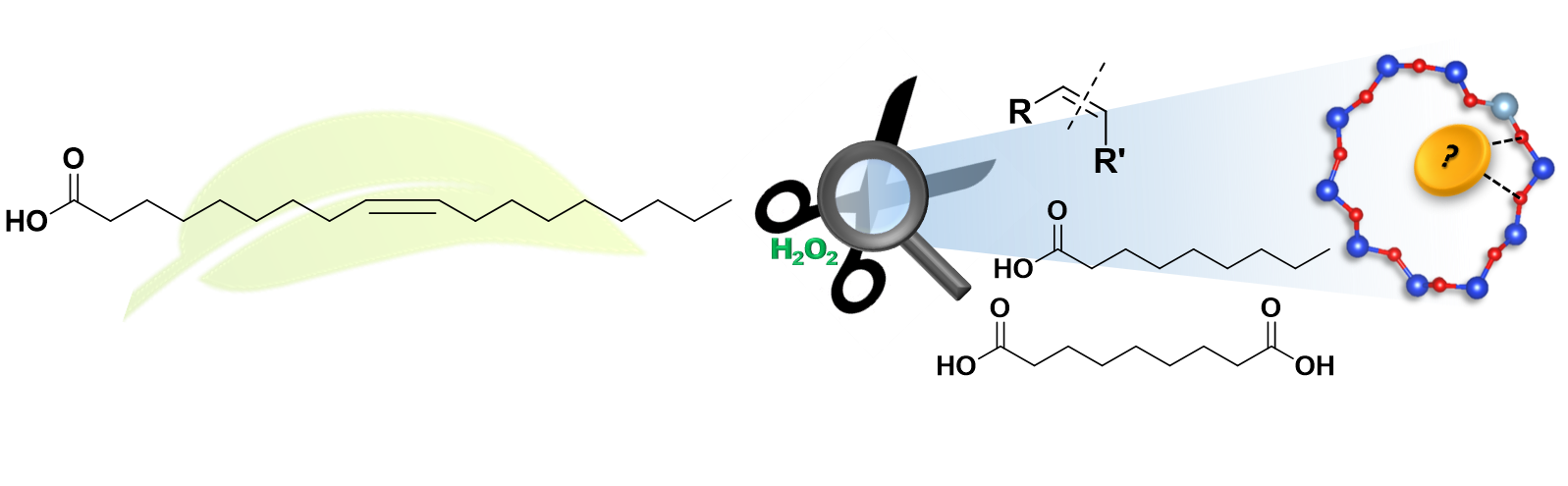
Catalytic Scissors: Oxidative Cleavage of C-C Bonds using Zeolite Catalysts
Formation and splitting of carbon-carbon bonds are one of the most essential chemical transformations, still rapidly growing in importance due to efforts to maximize the valorization potential of biomass into fine chemicals. The oxidative cleavage of C-C double bonds is a versatile chemical transformation, which allows for valorization of fatty acids, terpenes, lignin monomers or other bio-derived alkenes, yielding aldehydes and carboxylic acid with a variety of applications, suitable for a sustainable chemical industry. To come closer to this vision, we synthesize, characterize and test highly active and well-defined metal loaded zeolite catalysts, to efficiently catalyze the oxidative cleavage under mild conditions, aiming to fulfill all criteria of green chemistry.
PhD student: P. Treu
MSc thesis and HiWi jobs available
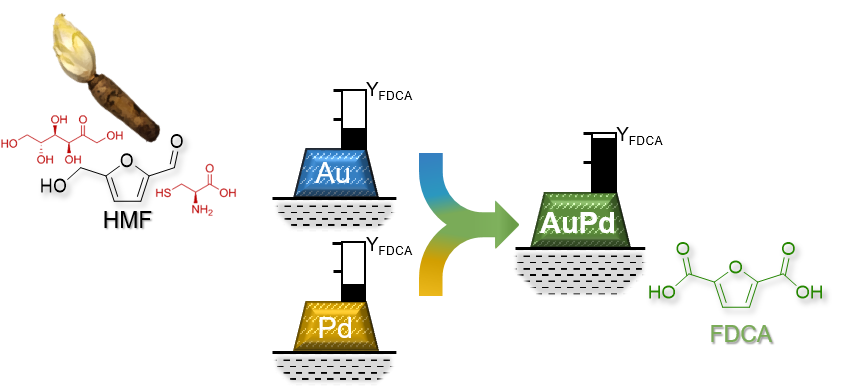
Knowledge-based Catalyst design for Valorization of Bio-based HMF
The production of bio-based monomers is a constantly growing field in industry and academia. The synthesis of 2,5-furandicarboxylic acid (FDCA) from 5-(hydroxymethyl)furfural (HMF) is particularly interesting due to the high quantity of possible bio-based feedstocks for HMF production, like cellulose or inulin. The state-of-the-art heterogeneous catalysts for this oxidation process are based on noble metals. However, the use of biomass for the production of FDCA is still a challenge due to its complex nature. To decrease costs and increase the sustainability of FDCA production, all stages of its production have to be optimized with minimal intermediate purification steps and biomass waste, e.g. chicory roots, should be used. To accomplish this, we study the influence of by-products in bio-based solutions on the catalysts and optimize the catalytic system accordingly. We aim to develop an efficient process for FDCA production by improving the reaction conditions and process design.
PhD student: D. Neukum
MSc thesis and HiWi jobs available
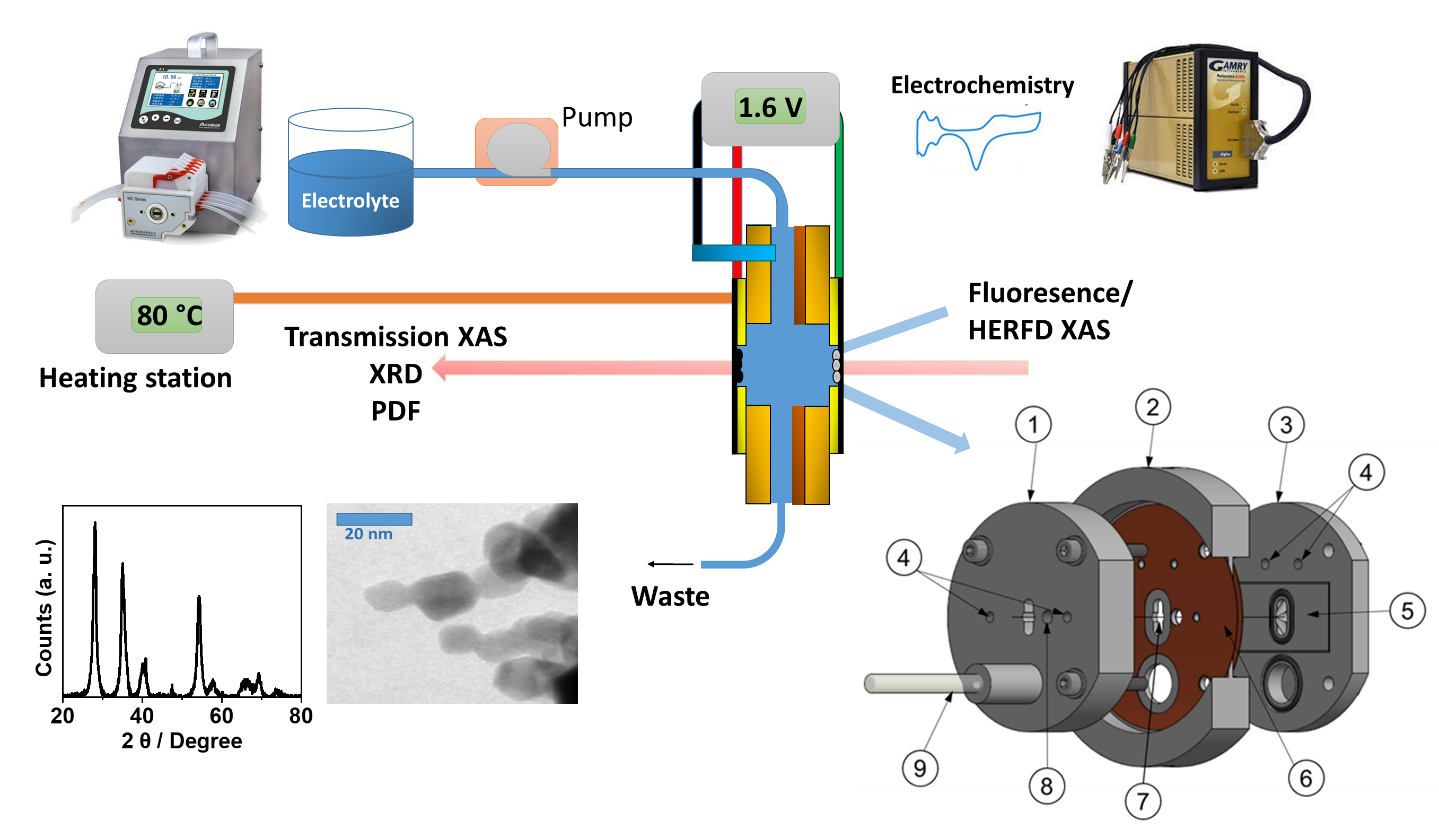
Operando investigation of OER for Green Hydrogen
The investigation of electrochemical catalysts by operando X-ray absorption spectroscopy (XAS) is essential to understand the processes occurring at the material-electrolyte interface under reaction conditions, with the goal to subsequently improve the catalyst. Electrochemical reactions of high interest are, for instance, water splitting (OER, HER) for the production of green hydrogen, and the CO2 reduction reaction for production of high-value chemical products. However, under industrially relevant conditions with high current densities, dynamic potential changes and elevated temperatures, operando XAS measurements become extremely challenging, due to the strong O2 bubble evolution on the catalyst surface during water splitting. We approach this challenge by a combining advanced electrochemical flow cells, refined spectroscopic approaches and advanced data analysis. The scope is to unravel the structural changes and the mechanism even under strong bubble evolution at high potentials of up to 1.6 V and temperatures of up to 80 °C.
Scientist: Dr. S. Czioska
MSc thesis and HiWi available
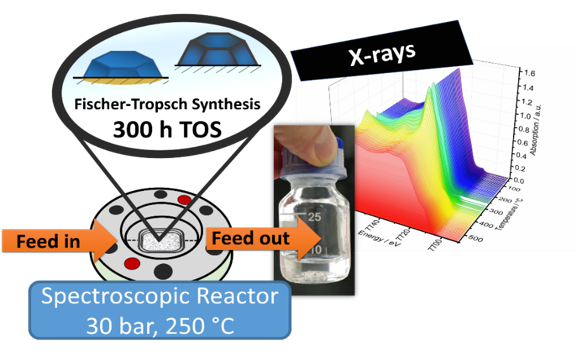
Sustainable Aviation Fuel
Aviation accounts for approximately 3% of global CO2 emissions, with projected increase by 2-folds in the next 15 years. Sustainable aviation fuels (SAF), unlike conventional fossil fuels, are based on green hydrogen and atmospheric CO2 and can be obtained via the Fischer-Tropsch-Synthesis technology (FTS). With the help of optimized FTS catalysts, SAF can be produced more efficiently. Their "drop-in" implementation can significantly reduce greenhouse gas emissions in industries such as aviation, where conventional fuels are difficult to replace. Catalyst design and optimization are crucial for advancing in the efficient production of SAF. In this context, we focus our research to preparation of model catalysts as well as operando characterization of FTS catalysts with the aim to understand structural properties and deactivation phenomena. For this operando XAS high-pressure and high-temperature cell reactors are developed.
PhD student: R. Elbuga-Ilica
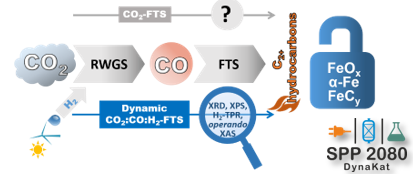
CO2 - FTS
CO2 hydrogenation into C2+-hydrocarbons using the Fischer-Tropsch synthesis (FTS) technology is an attractive way to mitigate the green-house effect and a highly important research topic in our group. Typically, CO2-FTS can be divided into two steps: (i) reverse water gas shift (RWGS) reaction to generate CO and (ii) sub-
sequent classical FT synthesis with CO. Fe-based materials are suitable candidates for this transformation. The on-going research focuses on developing catalysts with low methane selectivity but high selectivity to light (C2-C4) olefins and fuels. Alkali metal can be used as promoters to drive the selectivity to the desired products and improve the olefins/paraffins (O/P) ratio. However, the fundamentals of promoter-related improvements are still unclear. In collaboration with the group of E.V. Kondratenko from LIKAT (Rostock, DE) we aim at understanding the role of alkali promotors on Fe-based catalysts for the CO2-FTS using operando characterization.
Scientist: Dr. E. Saraçi
MSc thesis available