This research project is centered on a special class of polymers: the hydrogels. Not only do they contain hydrophilic groups but are also crosslinked and form a three dimensional network. Therefore they absorb water like a sponge; only much more efficient. They can take up 100g of water per 1 g of polymer or
|
|
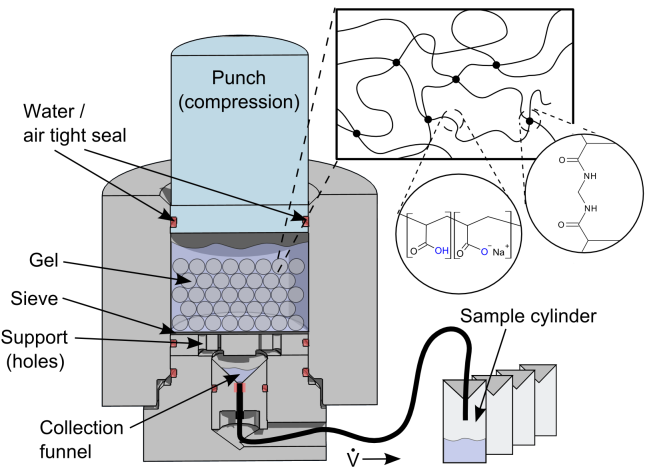
| Figure 1: Schematic, simple setup for the desalination using mechanical pressure. |
even more. These “super-absorbers” are widely used in diapers and similar applications. We are now applying these versatile materials to a new problem: the desalination of water. Sea water could be used for drinking or in agriculture, if it was not for the high content of dissolved salts (35 g/L, mostly sodium chloride). To separate the ions and the water is of great interest, as water is scarce in many regions. Already several techniques such as reverse osmosis or multistage flash distillation exist.
As the network already contains ionic groups that are fixed on the polymer strands, these charges will shield off the ions in the sea water. The hydrogel will absorb water containing much less salt. After separation from the low molecular ions is achieved, a second step is needed to regain the water from the polymer. We use a special inset (Figure 1) for a hydraulic press to apply the needed mechanical pressure to squeeze out the water, again like a sponge. The retained water would be, in the best case, fresh water.
We could already show that this idea does work in principle (Figure 2) and go on to explore some parameters that can be tuned to improve the process. This idea, as intriguing as it is, is still a long way from commercial application but a new concept, with possibilities we will continue to explore.
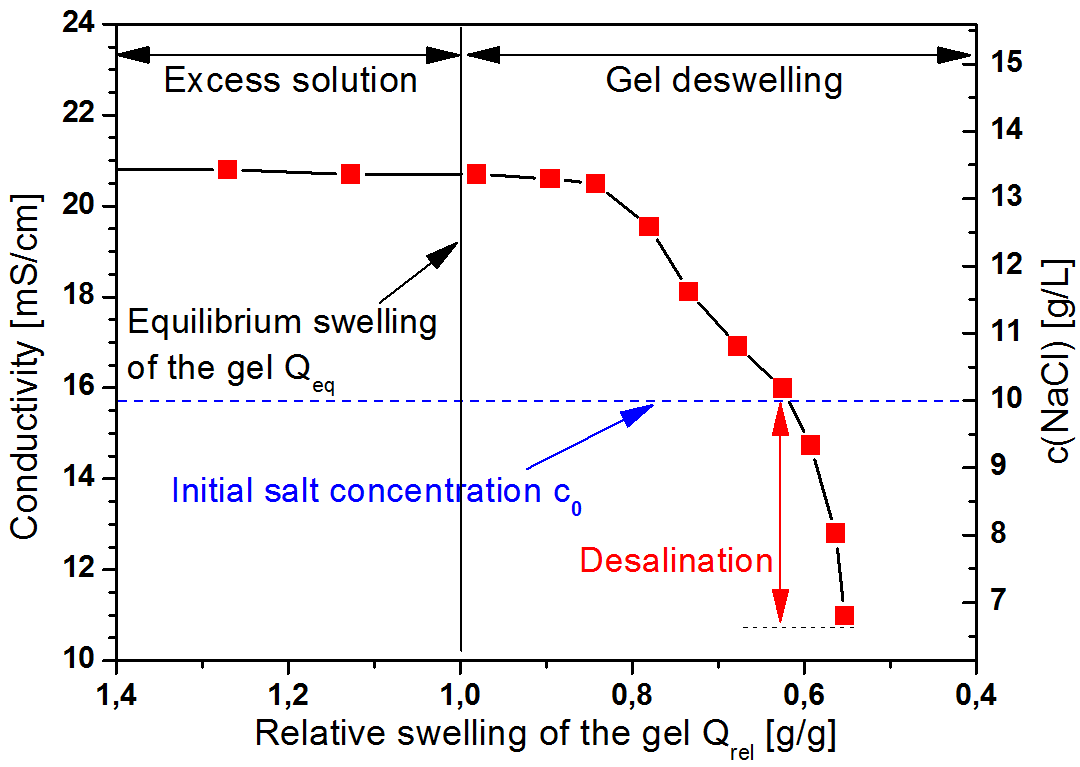
Figure 2: Typical result of a desaltination experiment with the lowest slat content at the end.
Other aspects of this project include the synthesis of model hydrogels and their characterization, especially their mechanical strength.
Further reading: J. Höpfner, C. Klein, M. Wilhelm, Macromol. Rapid Commun. 2010, 31, DOI 10.1002/marc.201000058 (online / in press).
Acknowledgements:The financial support of the Heinrich-Böll Foundation is gratefully acknowledged.